IDE Study
The IDE study, a prospective, multicenter, single-arm study, evaluated the safety of the Berlin Heart EXCOR® Pediatric VAD and compared its efficacy with a historical control population supported with ECMO as a bridge to cardiac transplantation in children. 48 patients of 16 years or younger were divided into two cohorts according to body-surface area: Cohort 1: n=24, BSA <0.7 m2, Cohort 2: n=24, BSA ≥0.7 to <1.5 m2
Results
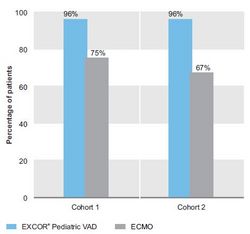
- The survival at 30 days in cohort 1 was 96% (46% transplanted, 50% on support) for the EXCOR® Pediatric VAD group and 75% (off ECMO) for the ECMO group.
- In cohort 2, the 30-day survival was 96% (21% transplanted, 4% weaned, 71% on support) for the EXCOR® Pediatric VAD group and 67% (off ECMO) for the ECMO group.
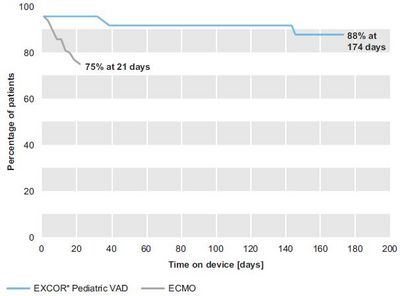
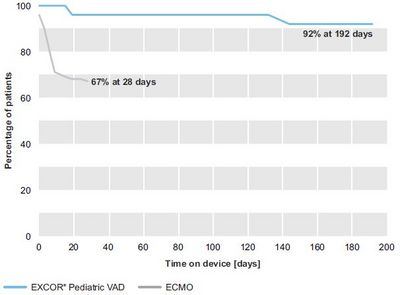
- In the EXCOR® Pediatric VAD group, all patients were off the device at 174 days in cohort 1 and 192 days in cohort 2, respectively. Overall, 88% in cohort 1 and 92% in cohort 2 were bridged to transplant or recovery with acceptable neurological outcome,respectively. In the ECMO group, for cohort 1 and 2 the longest time of support was 21 days and 28 days, respectively. 25% and 33% of the patients had died on ECMO support in cohort 1 and 2, respectively.
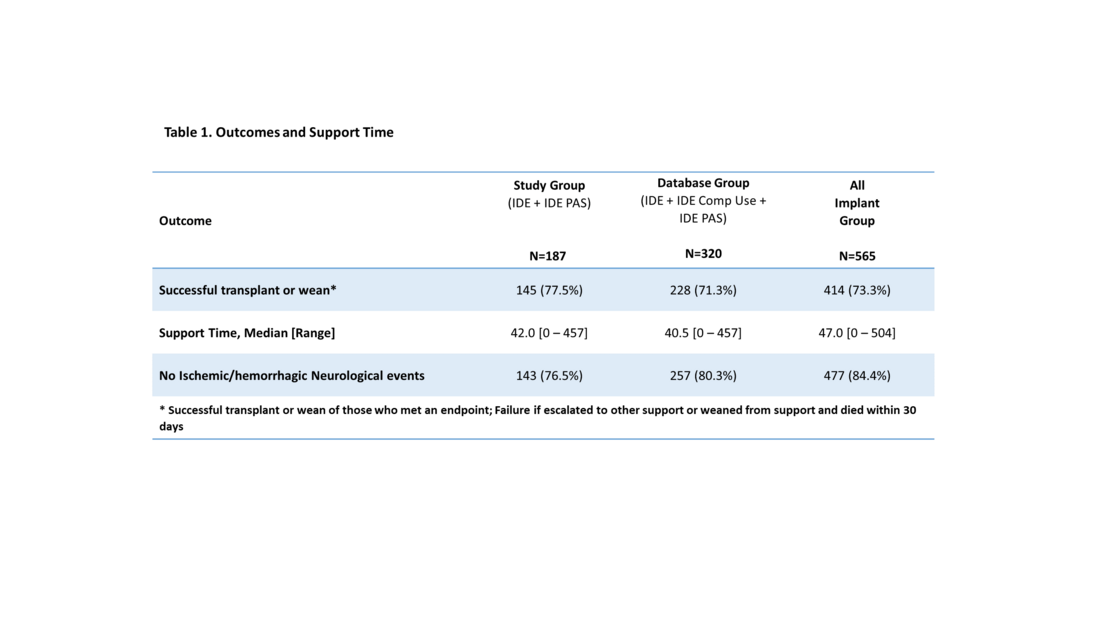
Berlin Heart received HDE approval by the U.S. Food and Drug Administration (FDA) for the EXCOR Pediatric in December 2011. As the sole condition of approval, a Post Approval study was conducted which enrolled 39 subjects. The final study report was submitted to FDA in February 2017.